製品
自己潤滑性、疲労特性、耐油性に優れます。歯車や軸受、自動車内装、医療部品等に採用されています。
様々な医療・ヘルスケア用品の部品としてPOMなどの樹脂材料が使用されています。
POMなどの樹脂材料は金属やガラスと比べて軽く、形状の自由度が高いという利点があります。
旭化成は、医療機器向けの樹脂材料として、POM(ポリアセタール)樹脂テナック™ をご提案します。
ポリアセタール樹脂(Polyoxymethylene 略称POM)は、強度・剛性、耐衝撃性、低摩耗、低摩擦、バネ回復性、耐クリープ性など、医療機器用途に最適な性質を有しています。特に様々な機構部品において、スムーズで静かな動作が期待できます。
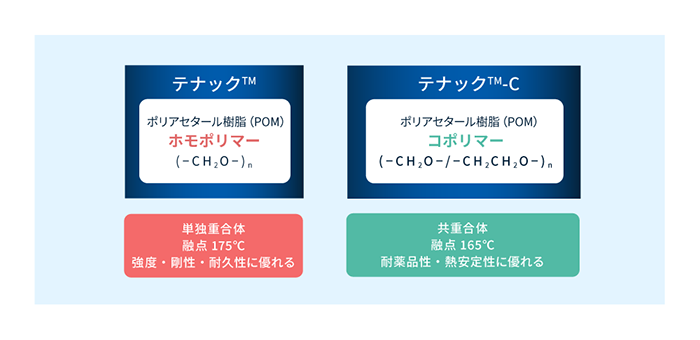
POMは主として(-CH2O-)の構造からなる熱可塑性樹脂です。
一般に(-CH2O-)の構造のみからなり、強度・剛性に優れるホモポリマーと、一部に(-CH2CH2O-)を含み、熱安定性に優れるコポリマーに大別されます。
旭化成は世界で唯一*ホモポリマー、TENAC™コポリマーの両方を生産しており、TENAC™用途に応じた供給が可能です。
*出典:富士経済「2025年エンプラ市場の展望とグローバル戦略」より。
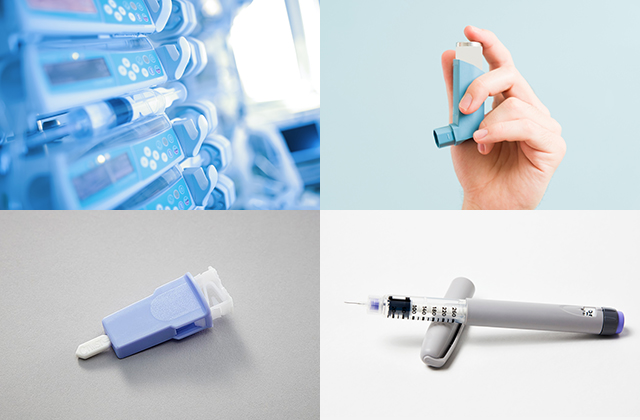
TENAC™医療機器向けPOM樹脂として、良成形性・良穿刺性(バネ特性)が求められるランセットや、高い耐摩擦摩耗性が求められる輸液ポンプガイドで採用実績があります。
医療機器向けPOM樹脂として新たに、テナック™ Q5010をラインナップしました。
テナック™ 医療用POM Q5010は中粘度のホモポリマーです。
医療用POMホモポリマー テナック™ Q5010規制・規格対応
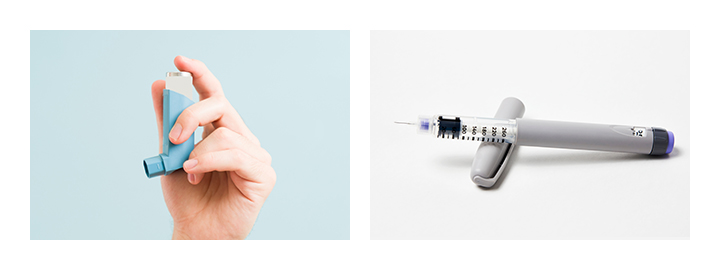
医療用POMホモポリマー テナック™ Q5010の用途として想定される医療機器のデバイスは、インスリンペンや喘息薬用吸入器の機構部品、バルブ部品、ギア類などです。
*次の用途には使用しないでください。体内、粘膜液血薬等に触れる医療容器・包装用具部品。食品、飲料水等に触れる容器・包装・器具・部品。