Product
Excellent heat resistance, strength and toughness, insulation, and oil resistance. It is widely used in automotive parts, electrical and electronic parts.
For inquiries regarding SDS and various chemical substance investigations, please make your request through your purchasing route, such as via a trading company.
We appreciate your understanding and cooperation.

There are two types of migration, electromigration and ion migration (electrochemical migration), and this page covers information on the latter, which occurs due to external factors.
Ion migration occurs when there is poor insulation between electrodes due to factors such as chemicals or heat, causing elution of the electrode metal and a subsequent short circuit.
To support our customers’ initiatives in making products used in electric and electronic applications, we offer a variety of engineering plastics that inhibit ion migration, haveare flame retardancet, and are superior in terms of various electrical properties (tracking resistance (CTI), etc.), glow wire ignition temperature (GWIT, etc.), long-term heat resistance (UL 746B RTI, etc.), and weather resistance, (UL 746C f1, f2).
For example, using a material that does not result in ion migration suppresses short circuits between terminals, contributing to improved product safety, and smaller, more compact product designs.
Asahi Kasei offers a range of products that are suitable for use in electric and electronic applications including flame retardant resin materials such as LEONA™ and XYRON™ , which eliminate ion migration triggered by flame retardants or slow progression compared to products that use red phosphorous.
This section covers testing methods for evaluating the ion migration resistance of resin materials.
As shown in Figure 1, copper electrodes are affixed to plates of various resin materials and high voltage is applied under high temperature and high humidity conditions.
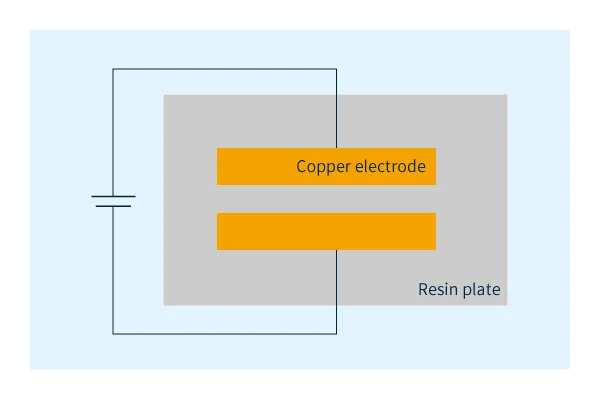
Ion migration occurs in the following way in this test:
1:
The flame retardant breaks down under high temperature, high humidity conditions, forming corrosive substances
2:
Copper electrodes dissolve, generating copper ions
3:
Copper ions receive an electron and are deposited as a metal
4:
Steps 1 to 3 above are repeated, and the deposited copper is gradually extended to the other electrode
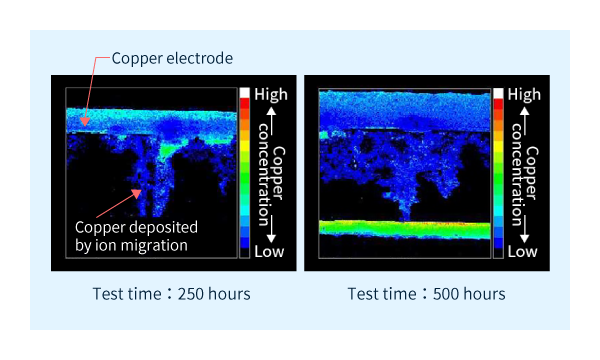
After exposure to the above testing conditions for a certain period of time, the copper distributed across the electrodes was researched using elemental analysis. When ion migration occurs, the copper element is gradually extended from one electrode to the other as shown in Figure 2.

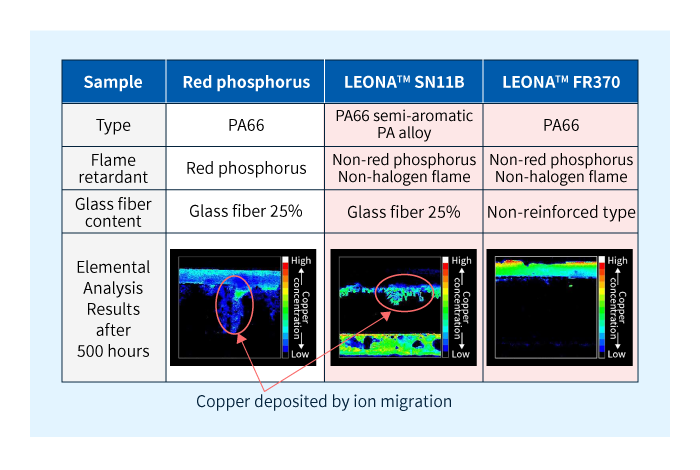
Next, the evaluation results for the LEONA™ flame retardant series are shown in Figure 4.
It was confirmed that there is no ion migration with the FR370. Although ion migration was confirmed to have occurred in the SN11B, the extension of copper was less than half that of the general product made using red phosphorus, confirming its slow progres
Please feel free to contact us with any questions about our products or technologies or to request samples.
We will introduce Asahi Kasei 's engineering plastic products and technologies in more detail.
We regularly deliver product and industry information to help you gather information.